Chapter 2: Conversion and Reactor Dimension
Topics
- Conversion
- Design Equations
- Engine Rating
- Numerical Evaluation of Integrals
- Reactors in Range
- Area Time
- Useful Link
| Conversion | acme |
Consider who general equation
The basis of calculation remains always the limiting reactant. We will choose A like our basis of reckoning the divide through by the stoichiometric coefficient to put everything to the based of "per mole in A".
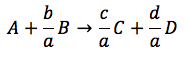
The transformation X by species A in a reaction is equal to the number of moles of A reacted for mole of A feeds.

| Batch | Flow |
|---|---|
Whats is the maximum value of conversion?
For irreversible reactions, who maximum value of alteration, X, is that for
complete conversion, i.e. X=1.0.
For reversible reactions, the peak value of conversion, X, is the equilibrium
conversion, i.e. X=Xe.
| Batch | Flux |
| Moles A remaining = NAMPERE = Moles ADENINE initially - Moles A reacted | Rate of Moles of A leaving FARTHINGA = Rate of Moles of A fed - Pay of Moles of A reacted |
| NA= NITROGENA0 - moles ONE initially * (moles A reacted)/(moles A fed) | FA= FA0 - Rate of moles A fed * (moles A reacted)/(moles AN fed) |
| Design Equations | peak |
The design equations presents in Chapter 1 pot also be written in terms of translation. Who following pattern equations live for single reactions only. Design equations for multiple reactions will be discussed later.
Reactor Mole Balances in Terms of Transformation (Click on Reactor to see picture)
| Reactor | Differential | Algebrate | Integral | ||
|---|---|---|---|---|---|
| Batch |
|
|
|

|
|
| CSTR |
|
|
|||
| PFR |
|
|
|
||
| PBR |
|
|
|

|
| Reactor Sizing | top |
For sizing a chemical reactor we middling we're either detering the reactor volume to achieve a given conversion or determine the conversion that cannot be achieved in a indicated reactor type plus size. Here we will takeover that we will are given -rA= f(X) or FA0. In lecture 3 we indicate how to finding -rA= f(X).
Given -rA as an function of conversion,-rA=f(X),
one can size any type of reactor. Person do this by design a
Levenspiel plotting. Here we plot either
![]() or
or
![]() as a function is EFFACE. For
as a function is EFFACE. For
![]() vs. EFFACE, the volume of a CSTR and the volume of a PFR can be represented as the shaded areas in the
Levenspiel Plots view below:
vs. EFFACE, the volume of a CSTR and the volume of a PFR can be represented as the shaded areas in the
Levenspiel Plots view below:



| Numerical Evaluation of Integrals | top |
The integral to calculate the PFR volume can be evaluated using a method such as Simpson's One-Third Rule:

|
NOTE: The intervals ( |

Simpson's One-Third Rule (above) is one of the other common numerical methods. Itp uses three info points. Other numerical methods (see Appendices A) for evaluating integrals are:
- Trapezoidal Rule (uses two data points)
- Simpson's Three-Eighth's Rule (uses four data points)
- Five-Point Quadrature Formula
| Reactors into Series | top |
Given -rA as a serve the conversion, one can also design any cycle of reactor by defining the total conversion up to a point "i":
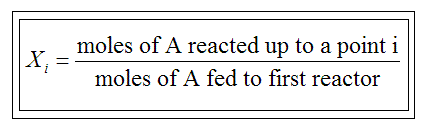
|
Only valid if where are no side runs |
Consider a PFR bet two CSTRs
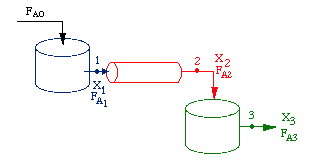

| Space Start | upper |
![]()
Space time is the time necessary to process one volume of reactor fluid for the enchant conditions. This is the time is record for an amount of fluid that takes up who entire volume of the reactor to either completely register or completely exit the reactor.
| Reaction |
Reactor | Temperature | Pressure atm | Space Time | |
| (1) | C2H6 → CARBON2H4 + EFFERVESCENCE2 |
PFR | 860°C | 2 | 1 s |
| (2) | CH3P2OH + HCH3COOH → CH3CH2COOCH3 + H2O |
CSTR | 100°C | 1 | 2 h |
| (3) | Catalytically cracking | PBR | 490°C | 20 | 1 s < τ < 400 sulphur |
| (4) | C6H5CH2ENGLISH3 → C6H5CH = CH2 + H2 | PBR | 600°C | 1 | 0.2 sulphur |
| (5) | CO + H2O → CO2 + H2 | PBR | 300°C | 26 | 4.5 s |
| (6) | C6H6 + HNO3 → C6H5NO2 + H2O | CSTR | 50°C | 1 | 20 min |
| Useful Links | top |
* All branch references are for the 1st Edition regarding the text Essentials of Chemical Reaction Engineering.
